Abstract
Background
During resistance training, quercetin ingestion can enhance motor unit (MU) with a higher recruitment threshold in older adults.
Objective
We investigated the effects of daily quercetin glucoside ingestion on chronic adaptations in muscle strength and MU behavior following resistance training in healthy older adults.
Methods
Twenty-six older adults were randomly allocated to two groups that completed 6-week resistance training intervention with the ingestion of either placebo (PLA) or quercetin glycosides (QUE) at 200 mg/day. Maximal voluntary force (MVF) during isometric knee extension, muscle mass, and MU firing behavior during ramp task at 70%MVF were measured before (PRE) and after (POST) intervention.
Results
In both groups, knee extensor MVF was significantly increased (both p < 0.001), and the improvement in QUE (115.1 ± 11.0%) was greater than in PLA (105.3 ± 4.8%) (p < 0.001) by the Mann–Whitney test. Muscle mass was not changed from PRE to POST in PLA or QUE (p > 0.050). At POST, firing rates of Mus with relatively moderate (recruited between 20 and 40%MVF) or higher (recruited between 20 and 40%MVF) recruitment thresholds were higher in QUE than PLA (p < 0.050). There was a significant correlation between %change in MVF and %change in firing rates of MUs with a relatively higher recruitment threshold from PRE to POST (p = 0.018, r = 0.642).
Conclusions
These results suggest that the adaptations of MUs with higher recruitment thresholds explain the greater improvement in muscle strength associated with QUE ingestion.
Clinical trial registry
UMIN000053019 (https://rctportal.niph.go.jp/detail/um?trial_id=UMIN000053019).
Introduction
Aging-related loss of muscle strength, which is attributed to changes in motor units (MU), such as myofiber atrophy [1] and denervation [2], is associated with increased risks of falls and fractures, physical disability, loss of independence, lifestyle-related diseases, and mortality [3, 4]. Resistance training has been widely recognized as a method to offset some age-related loss of muscle strength [5,6,7,8].
Previous meta-analyses reported that high-intensity resistance training has the greatest effects on muscle strength in comparison with moderate or low-intensity training in older adults [9, 10]. One of the factors determining the effects of resistance training on muscle strength may be the difference in behavior of MUs with a higher recruitment threshold during such training. Increasing training intensity leads to the activation of MUs with a higher recruitment threshold [11]. According to the functional properties associated with the motor neuron size, MUs with a higher recruitment threshold are related to type II muscle fibers [12, 13]. Muscle atrophy is observed with aging, in which type II muscle fibers are more affected than type I fibers [2].
However, older people (i.e., frail elderly, unskilled resistance-exercise practitioners, and people with joint or cardiorespiratory problems) may not be able to cope with the significant load involved in this moderate-to-high-intensity training [14, 15]. A previous review of 121 studies on resistance training in older adults showed that adverse events are more frequently reported in research on older adults involving studies implementing high-intensity resistance training [14]. Therefore, high-intensity resistance training can recruit MUs with a higher threshold during training, but there are safety issues for older adults. Hence, methods for activating MUs with a higher recruitment threshold other than increasing intensity may be effective to minimize age-related muscle strength loss. Whole-body vibration decreased the recruitment threshold of MU with a higher recruitment threshold [16], and blood flow restriction induced recruitment of MUs with a higher recruitment threshold [17]. These methods enhance the increase in muscle strength when combined with resistance training in older adults [18, 19]. These data suggest that methods which activate MUs with a higher recruitment threshold during a single session of resistance exercise may constitute an effective strategy to minimize age-related muscle strength loss. However, since these methods require specialized equipment and administration, simpler methods of activating MUs with a higher recruitment threshold should also be examined.
Quercetin is structurally similar to adenosine; it blocks adenosine-binding to A1 and A2a receptors and releases neurotransmitters such as acetylcholine and dopamine [20], and quercetin ingestion has been shown to alter MU firing patterns [21,22,23]. Previous studies revealed that the recruitment threshold was lowered and firing rate was increased in MU with a higher recruitment threshold by single or continuous quercetin ingestion, evaluated using high-density surface electromyography (HDsEMG) [21,22,23]. Also, previous studies showed that a single ingestion of quercetin increased the median frequency of surface EMG [24], and reported that 2-week quercetin ingestion increased muscle fiber conduction velocity [25]. These findings also suggest that quercetin ingestion lowers the overall recruitment threshold of MUs and activates MUs with a higher recruitment threshold. Hence, its ingestion may enhance the effects of resistance training on muscle strength in older adults.
This study aimed to investigate the effect of daily quercetin ingestion on MU behavior during a single session of resistance training and adaptations to chronic resistance training. We hypothesized that quercetin ingestion activates MUs with a higher recruitment threshold following a single session of resistance training. Additionally, we hypothesized that quercetin ingestion enhances the increase in muscle strength by altering the adaptation of MUs with higher recruitment thresholds following resistance training.
Methods
Participants
Thirty healthy older male and female adults, aged 65–82 years, participated in this study. The participants were physically independent and in good health. Inclusion criteria were as follows: 65 years old and over; no acute disease; body mass index (BMI) between 18.5 and 35 kg/m2; without any exercise restrictions imposed by medical doctors. Exclusion criteria included dependence on walking aids, the presence of artificial joints or who regularly consume foods with functional claims (government-approved foods specified health uses) containing high levels of quercetin. Participants were instructed to refrain from the ingestion of other possible ergogenic aids sold as foods with functional claims during the intervention. All participants provided written informed consent, the research ethics committee of Chukyo University approved the study protocol (approval number: 2022-004), and it was conducted in accordance with the Declaration of Helsinki. The present study was registered in the University Hospital Medical Information Network Clinical Trial Registry (UMIN000053019). Participants with metabolic disease, upper, acute infection, and those under medications that impact muscle protein metabolism and neural activity were excluded. All data were collected at Chukyo University.
Study design
Resistance training
All participants trained their knee extensor muscles for 6 weeks (Fig. 1). We applied isometric knee extension resistance training on both legs three times a week. Participants were seated on a chair with their ankle fixed on a force transducer and performed three sets of 10 isometric contractions during unilateral knee extension for both left and right legs with a rest interval of 120 s. Both legs were trained in separate sessions. The intensity was defined as 60% of maximal voluntary force (MVF) and the performed force could be monitored in real-time on a screen via a transducer. MVF used in the training were based on MVF at the pre-intervention. One contraction comprised a 5-s rest phase and 5-s sustained phase at or exceeding the target torque. During contractions, the experimenter requested that the participants count aloud for 5 s to maintain breathing.
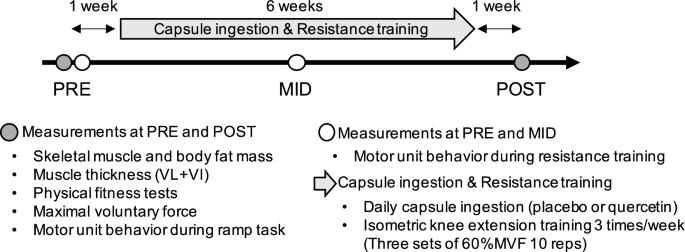
Intervention overview. PRE, before intervention; MID, 3 weeks after start of capsule ingestion and resistance training intervention; POST, after intervention; VL, vastus lateralis; VI, vastus intermedius; MVF, maximal voluntary force
Supplementation
During this resistance training intervention, participants were randomly allocated to placebo (PLA) or quercetin (QUE) groups (PLA, Age: 73 ± 4 years, Height 157.9 ± 7.2 cm; Body mass: 54.3 ± 5.8 kg; QUE, Age: 72 ± 5 years, Height: 160.3 ± 7.1; Body mass: 58.6 ± 10.9 kg). Randomization was performed based on dynamic allocation to maintain balance among the groups in age, sex, and muscle strength during familiarization of measurement for isometric knee extension strength using a spreadsheet program with the RAND function (Microsoft Excel 2021, Microsoft Corporation, Redmond, WA, United States). The participants took two gelatin-coated and colored capsules containing 200 mg of quercetin glycosides with 1.8 g of dextrin (quercetin) or 2.0 g of dextrin (placebo), which were not any difference visually and olfactory. A randomized, double-blind, placebo-controlled treatment was conducted for capsule intervention. The randomization codes for both participants and groups were held in sealed opaque envelopes by three different individuals who were not engaged in the present study and were kept securely in a locked vault. After the analysis was completed, the safe was unlocked and the envelope was opened. Quercetin glycosides are more water-soluble and bioavailable than quercetin aglycone, which does not exist in a glycosidic or conjugated form [26]. When absorbed, quercetin glycosides are enzymatically converted into the aglycone form and exhibit beneficial effects similar to those of the corresponding quercetin aglycone. Quercetin glycosides were enzymatically manufactured at San-Ei Gen F.F.I., Inc. (Osaka, Japan) from isoquercitrin prepared from quercetin-3-O-rutinoside. In a GRAS statement from FDA, up to 293 mg/serving of quercetin is acceptable. The participants took the capsules with drinking water every morning. A dietary survey was performed by a nutritionist with a license from the Ministry of Health, Labour and Welfare, Japan, using a brief-type self-administered diet history questionnaire (BDHQ) [27] to quantify total energy, carbohydrates, protein, and fat in daily diets, and values were normalized by individual body mass in order to estimate participants’ nutritional conditions during intervention. This questionnaire was applied at the time of intervention, and so the results should reflect nutritional conditions during that period. The participants’ total physical activity during intervention was evaluated utilizing the short-form International Physical Activity Questionnaire (IPAQ) and calculated as the sum of metabolic equivalents (MET) × minute per week for occupation, transportation, housework, and recreational activity.
Measurements
The participants underwent 6-week resistance training and capsule ingestion intervention (Fig. 1). Measurements of muscle strength and the MU firing pattern, anthropometry tests, and physical fitness tests were conducted before (PRE) and after (POST) intervention. There was a 1 week washout period between measurements and intervention. Additionally, MUs behavior during resistance training was assessed on the same day as the PRE measurements and again three weeks after the start of the intervention (MID). Before PRE, participants familiarized themselves with the measurements and motor tasks. Participants were instructed to refrain from vigorous-intensity exercise and the consumption of foods or drinks containing quercetin or caffeine 24 h prior to testing [28].
The primary outcome in the present study was MVF as muscle strength. Secondary outcomes were mucle thickness, muscle mass, physical fitness tests, and motor unit activity, and others were height, body mass, fat mass, nutritional parameters and physical activity.
Measurements before and after intervention
Body composition
First, height was measured in participants not wearing shoes by stadiometer (Seca 213, seca Nihon, Chiba, Japan) and body mass was measured, skeletal muscle mass and body fat mass were estimated using a bioelectrical impedance analysis device (Inbody 430, Inbody Japan Inc., Tokyo, Japan). The device assesses the resistance and reactance of the arms, trunk, and legs with a segmental multi-frequency approach (5, 50, and 250 kHz). Muscle volume of the lower right limb was evaluated to examine the impact of intervention.
Muscle thickness
The participants sat on a chair and their knee was fixed at 90-degree flexion in a resting position. Since the dominant foot is right in all participants, muscle thickness and MUs behavior measurements were conducted on the right foot. Ultrasound images of vastus lateralis and vastus intermedius muscles of the right leg were obtained using an ultrasound device (LOGIQ e Premium, GE Healthcare) with a 10-MHz linear array probe. The measurement location was determined as 50% of the distance from the head of the great trochanter to superior lateral edge of the patella. Two longitudinal images were acquired while paying attention to placing the probe with minimal pressure and adjusting its angle so that the bone echo was brightest and fascia- and bone-muscle boundaries were parallel. Muscle thickness was measured as the distance between the superficial and bone-muscle boundaries, determined as the mean value between two longitudinal images.
Physical fitness tests
Physical fitness tests comprised the Timed Up and Go (TUG) and chair stand tests. Participants were given verbal instructions to stand up from a chair, walk 3 m, cross a mark on the floor, turn around, walk back, and sit down on the same chair. They performed two TUG tests: one where they walked at their normal speed (TUGnormal) and one where they walked as fast as they could (TUGfast). The time taken to stand up from the chair, complete the task and sit down again was measured. After TUG, they were asked to stand up from and sit down on the chair as fast as possible within 30 s with their arms folded across their chests [29]. The time required to stand up and sit down 5 times (Chair5times) and number of times they could stand up and sit down within 30 s (Chair30sec) were also measured. All physical fitness tests were performed using the same chair and with similar ambient conditions. The chair was of a standard height (0.43 m) without armrests.
Maximal voluntary force
MVF during isometric knee extension for the right leg was measured by a custom-made dynamometer (Takei Scientific Instruments Co., Ltd., Niigata, Japan) with a force transducer (LU-100KSE; Kyowa Electronic Instruments, Tokyo, Japan). The participants sat in the dynamometer with both hip and knee joint angles set to 90°and the distal part of the shank of their right leg fixed to the force transducer. After standardized warm-up protocols (submaximal isometric contraction of 2 × 50 and 2 × 80%), MVF of isometric knee extension was measured. MVF force was measured at least twice, and again if an increase of more than 5% was observed. MVF torque was calculated by multiplying the force and arm length, between the knee joint axis and force transducer.
High-density surface electromyography
After measuring MVF, participants performed a submaximal isometric knee extension task to record high-density surface electromyography (HDsEMG) (Details described below) to identify individual MUs behavior in the vastus lateralis muscle using the same dynamometer. They performed a ramp contraction task that consisted of a 14-s increasing phase to 70%MVF and 5-s sustained phase of 70%MVF at PRE and POST. The ramp task at PRE and POST was normalized by MVF at PRE. Real-time visual feedback for the force signals and target force were displayed on a monitor.
Measurement of motor unit behavior in resistance training before and during intervention
Participants performed three sets of ten isometric knee extensions executed at 60%MVF at PRE for 5 s with an inter-contraction interval of 5 s and inter-set interval of 120 s, which simulate a single session of resistance training. Real-time visual feedback for the force signals and target force were displayed on a monitor. MU firing rates during first and last contractions of resistance exercise were measured. Prior to resistance training, 70%MVF in the PRE ramp task was performed to determine the MU recruitment threshold.
Recording of high-density surface electromyography
HDsEMG signals were recorded from the vastus lateralis using a semi-disposable adhesive array of 64 electrodes, each with a diameter of 1 mm and spaced 8 mm apart (GR08MM1305, OT Bioelettronica, Turin, Italy). The electrodes were arranged in 13 rows and 5 columns, with one missing electrode in the distal left corner. The center of the electrode array was determined as the midpoint between the greater trochanter and superior lateral edge of the patella, and the columns were aligned along the line connecting these two points. A reference electrode (WS2, OT Bioelettronica, Turin, Italy) was affixed to the proximal end of the shank. The monopolar HDsEMG signals were filtered (10–500 Hz), amplified by a factor of 256, and digitized at a sampling rate of 2000 Hz through a 16-bit analog-to-digital converter (Sessantaquattro, OT Bioelettronica, Turin, Italy). The signal from the force transducer was synchronized with the recorded HDsEMG signals in the analog-to-digital conversion process. Signal processing and analysis were performed using MATLAB software (MATLAB R2019a, MathWorks GK, Tokyo, Japan). To identify individual MUs, we used the Convolution Kernel Compensation (CKC) method with DEMUSE software (ver. 5.0.1; University of Maribor, Slovenia) applied to monopolar surface electromyogram (EMG) data recorded during the experiment [30]. After decomposition, an experienced investigator reviewed the firing times of individual MUs. Based on these values, instantaneous MU firing rates were calculated. These procedures are consistent with those used in previous studies [21, 31, 32].
During the ramp contraction task measurement (PRE and POST), the individual MU firing rate was calculated as the mean value between 62.5 and 67.5%MVF, and the MU recruitment threshold was defined as the force level where the initial firing was identified [33, 34].
During resistance training (PRE and MID), to investigate acute MU activity responses following a single session of resistance exercise, we measured MU firing rates during the first and last contractions of resistance exercise. The mean of interspike intervals between MU spikes at 57.5 and 62.5%MVF was used to determine the MU firing rate [33, 34]. Prior to the resistance training session, the MU recruitment threshold was defined as the force level where the initial firing was identified during the ramp task. The detection of MUs was performed via the CKC technique to establish MU identification filters from HDsEMG signals collected during the ramp task and applied to HDsEMG signals obtained during resistance exercise [35]. This method has been demonstrated to efficiently track individual MU firing patterns across different times [22, 33, 36]. In other words, the recruitment threshold was measured by the ramp task and change in MU firing rate from the first to last contractions during a single session of resistance training. This allowed us to examine effects of fatigue due to resistance training on individual MUs with different recruitment thresholds [33]. To investigate the effects of quercetin and resistance training on different MU recruitment thresholds, MUs with such thresholds between 0 and 20%MVF, 20 and 40%MVF, 40 and 60%MVF, and all MUs were grouped into MU0–20, MU20–40, MU40–60, and MUALL, respectively, based on previous research [32, 37].
To investigate the details of the change in MUs behavior from before to after intervention, we performed MU tracking from PRE to POST using the method described above. This was achieved by concatenating the HDsEMG recordings from pre- and post ingestion, and then applying the individual motor unit filters to the entire concatenated dataset [38, 39]. The action potentials of MU that could be tracked from PRE to POST were confirmed, and in accordance with the criteria of a previous study [40], MUs with a correlation coefficient of > 0.7 were included in the analysis.
Statistics
The results are reported as the mean ± standard deviation. Power analysis was performed with an effects size of 0.96, the power set at 0.80, and an a-level of 0.05 under the hypothesis that strength gain following resistance training with quercetin ingestion is greater than without quercetin ingestion. Priori power analysis in G*power determined that 30 participants were required in this study. We calculated %change in MU firing rate from first to last contractions during a single session of resistance training as the MU firing rate during the first/last contractions, or %change in MVF from PRE to POST as MVF at POST/PRE. To analyze the relationship between adaptations of MVF and the MU firing rate, we averaged firing rates of MU0–20, MU20–40, MU40–60, and MUALL at PRE and POST for each participant and calculated %change in MU firing rate from PRE to POST as POST/PRE.
Dependent t-tests were used to determine whether there were any differences between groups in age, height, body mass, fat mass at PRE, nutritional parameters, neuromuscular properties or physical activity. For MVF, muscle thickness of the vastus lateralis and vastus intermedius, and muscle volume of the right lower limb were Levene test was performed to evaluate variance homogeneity between groups and the sphericity assumption for the repeated measures was examined with Mauchly’s test of sphericity. Then, analysis of covariance (ANCOVA) was performed, using the value at PRE as the covariate for MVF, muscle thickness of the vastus lateralis and vastus intermedius, and muscle volume of the right lower limb. Mixed 2-way (Group: PLA and QUE, Time: PRE and POST or PRE and MID) analysis of variance (ANOVA) was performed to assess the MU firing rate during ramp contraction and %change in MU firing rate from first to last contractions during resistance training. We compared %change in MVF from PRE to POST between PLA and QUE by the Mann–Whitney test. When appropriate, follow-up analyses were performed using dependent- and independent-sample t-tests with Bonferroni corrections. Because the number of MU that could be tracked between PRE and POST was limited, we used non-parametric statistical tests. The Wilcoxon test was used to compare firing rates of MU tracked among PRE and POST. Associations between %change in MVF and %change in MU firing rate from PRE to POST were analyzed by the Pearson correlation coefficient (r). Partial eta squared (η2) was used to quantify magnitude of effect size in ANOVA and ANCOVA where small ≥ 0.01, moderate ≥ 0.08, large ≥ 0.26, very large ≥ 0.50. The magnitude of effect size in t-test and post-hoc for ANOVA and ANCOVAwas quantified using Cohen d’s (d), with values classified as follows: small ≥ 0.10, moderate ≥ 0.30, large ≥ 0.50. Furthermore, r was used quantify magnitude of effect size in non-parametric analysis where small ≥ 0.10, moderate ≥ 0.30, large ≥ 0.50. The level of significance was set at α = 0.05, and all statistical analyses were performed using SPSS (version 25.0, SPSS, Tokyo, Japan).
Results
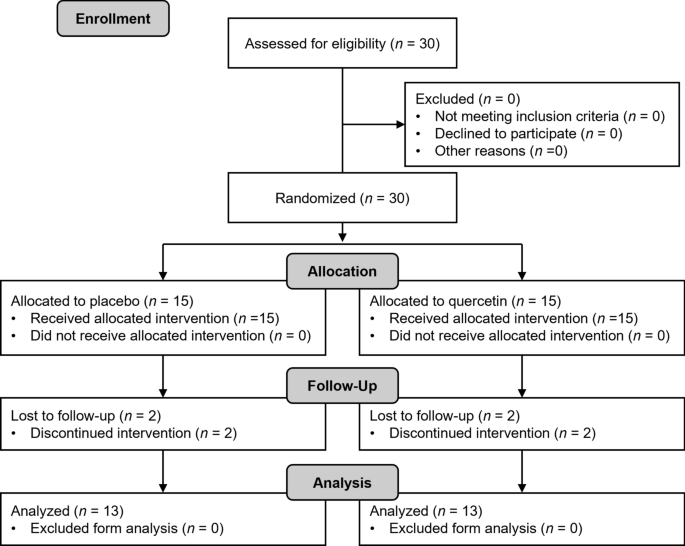
Six males and 7 females in QUE (Age: 73 ± 4 years) and 5 males and 8 females in PLA (Age: 71 ± 5 years) completed the intervention (Fig. 2). Four were excluded due to scheduling conflicts or other personal reasons. Self-reported supplement compliance was 99.7% and 99.9% in QUE and PLA. Resisance training adherence was 99.8% in PLA and QUE, respectively. There were no significant differences in the characteristics of the participants at PRE, or nutritional parameters and physical activity during the intervention between PLA and QUE (all, p > 0.050) (Table 1). Additinoally, MVF (p = 0.133,d = 0.23), TUGnormal (p = 0.520, d = 0.06), TUGfast (p = 0.677, d = 0.05), Chair5times (p = 0.340, d = 0.11), Chair30sec (p = 0.880, d = 0.08), muscle thickness (p = 0.633, d = 0.12), muscle mass (p = 0.230, d = 0.14) and firing rate of MUALL (p = 0.900, d = 0.03), MU0–20 (p = 0.777, d = 0.09), MU20–40 (p = 0.660, d = 0.03), MU40–60 (p = 0.890, d = 0.03) at PRE did not significant difference between PLA and QUE.
Flowchart of this study. Thirty participants recruited and randomly allocated placebo (7 males and 8 females) and quercetin (7 males and 8 females)
Motor unit behavior during a single session of resistance training
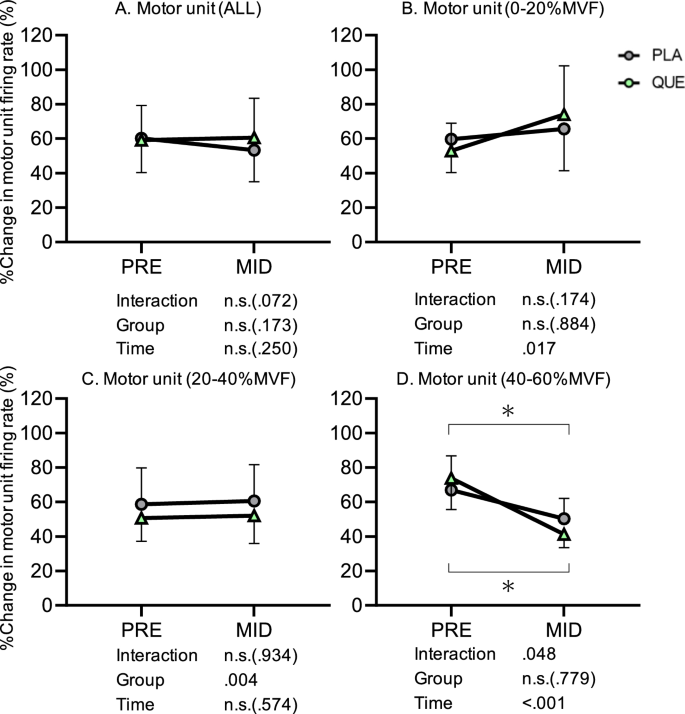
Numbers of detected MUALL for PLA and QUE were 85 and 87 in PRE and 64 and 71 in MID, respectively. Regarding %change in firing rate of MUALL from the first to last contractions, there was neither an interaction (Group x Time, p = 0.072, η2 = 0.09) nor main effect of Group (p = 0.173, η2 = 0.08) or Time (p = 0.250, η2 = 0.06) (Fig. 3A). Numbers of detected MU0–20 for PLA and QUE were 26 and 16 in PRE and 19 and 9 in MID, respectively. Concerning %change in firing rate of MU0–20 from first to last contractions, there was no interaction (Group x Time, p = 0.174, η2 = 0.01) nor main effect of Group (p = 0.884, η2 < 0.01), but there was a main effect of Time (p = 0.017, η2 = 0.15) (Fig. 3B). Numbers of detected MU20–40 for PLA and QUE were 44 and 45 in PRE and 33 and 45 in MID, respectively. For %change in firing rate of MU20–40 from first to last contractions, there was no interaction (Group x Time, p = 0.934, η2 < 0.01) or main effect of Time (p = 0.574, η2 = 0.02), but there was a main effect of Group (p = 0.004, η2 = 0.18) (Fig. 3C). Numbers of detected MU40–60 for PLA and QUE were 15 and 26 in PRE and 12 and 17 in MID, respectively. Regarding %change in firing rate of MU40–60 from first to last contractions, there was a significant interaction (Group x Time, p = 0.048, η2 = 0.13) and main effect of Time (p < 0.001, η2 = 0.28), but there was no main effect of Group (p = 0.779, η2 = 0.02) (Fig. 3D). Post-hoc tests revealed that %change in MU firing rate was significantly decreased from PRE to MID in PLA (p = 0.046, d = 0.30) and QUE (p < 0.001, d = 0.58).
%Change in firing rate from first to last contractions during a single session of resistance training of all motor units (A) and motor unit recruitment threshold between 0 and 20%MVF (B), 20 and 40%MVF (C), and 40 and 60%MVF (D) in placebo (PLA, gray) (5 males and 8 females) and quercetin (QUE, green) (6 males and 7 females) groups before (PRE) and after 3 weeks of (MID) intervention. *p < 0.050
Measurements before and after intervention
Maximal voluntary force and physical fitness tests
For MVF, there was an interaction (Group x Time, p = 0.036, η2 = 0.11) and main effect of Time (p < 0.001, η2 = 0.18), but there was no main effect of Group (p = 0.240, η2 = 0.08) (Table 2). Post-hoc tests revealed that MVF was significantly increased from PRE to POST in PLA and QUE (PLA: p < 0.001, d = 0.22; QUE: p < 0.001, d = 0.31) (Table 2). In QUE, %change in MVF from PRE to POST was significantly greater than in PLA (p < 0.001, r = 0.55) (Table 2).
For all physical fitness tests, there was no interaction (Group x Time) or main effect of Group or Time (Table 2).
Muscle thickness
For muscle thickness of the vastus lateralis and vastus intermedius, there was no interaction (Group x Time, p = 0.364, η2 = 0.04) or main effect of Group (p = 0.236, η2 = 0.05) or Time (p = 0.264, η2 = 0.05) (Table 2). Concerning muscle volume of the right lower limb, there was no interaction (Group x Time, p = 0.454, η2 = 0.03) or main effect of Group (p = 0.443, η2 = 0.03) or Time (p = 0.121, η2 = 0.04) (Table 2).
Motor unit behavior
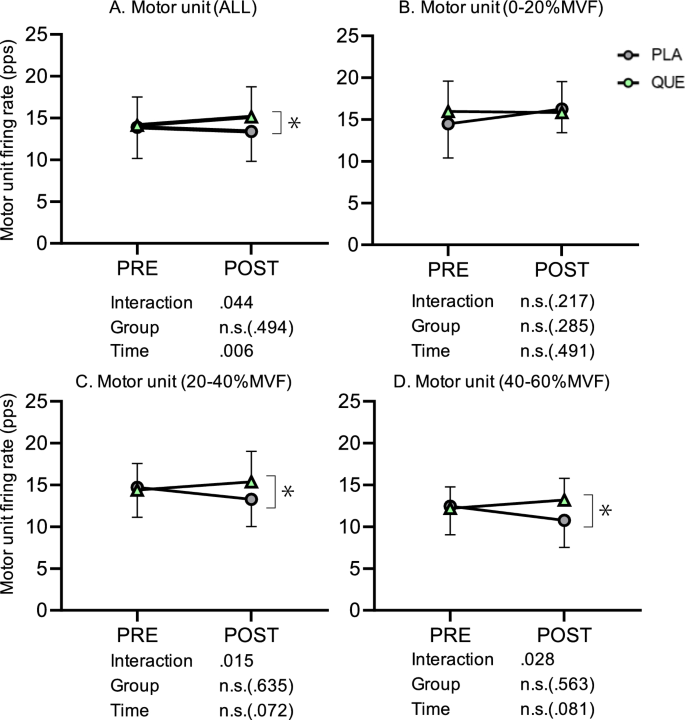
Numbers of detected MUALL in PLA and QUE were 65 and 99 in PRE and 112 and 135 in POST, respectively. Regarding the firing rate of MUALL, there was an interaction (Group x Time, p = 0.044, η2 = 0.11) and main effect of Time (p = 0.006, η2 = 0.09), but there was no main effect of Group (p = 0.494, η2 = 0.04) (Fig. 4A). Post-hoc tests revealed that the firing rate of MUALL was significantly greater in QUE than PLA in POST (p = 0.001, d = 0.19) (Fig. 4A). Numbers of detected MU0–20 in PLA and QUE were 13 and 22 in PRE and 27 and 37 in POST, respectively. For the firing rate of MU0–20, there was no interaction (Group x Time, p = 0.217, η2 = 0.06) or main effect of Group (p = 0.285, η2 = 0.05) or Time (p = 0.491, η2 = 0.04) (Fig. 4B). Numbers of detected MU20–40 in PLA and QUE were 29 and 50 in PRE and 61 and 75 in POST, respectively. For the firing rate of MU20–40, there was a significant interaction (Group x Time, p = 0.015, η2 = 0.22), but there was no main effect of Group (p = 0.635, η2 = 0.04) or Time (p = 0.072, η2 = 0.09) (Fig. 4C). Post-hoc tests revealed that the firing rate of MU20–40 was significantly greater in QUE than PLA in POST (p = 0.002, d = 0.60) (Fig. 4C). Numbers of detected MU40–60 in PLA and QUE were 23 and 27 in PRE and 24 and 23 in POST, respectively. Concerning the firing rate of MU40–60, there was a significant interaction (Group x Time, p = 0.028, η2 = 0.22), but there was no main effect of Group (p = 0.563, η2 = 0.09) or Time (p = 0.081, η2 = 0.11) (Fig. 4D). Post-hoc tests revealed that the firing rate of MU40–60 was significantly greater in QUE than PLA in POST (p = 0.038, d = 0.58) (Fig. 4D).
Firing rate of all motor units (A) and motor unit recruitment threshold between 0 and 20%MVF (B), 20 and 40%MVF (C), and 40 and 60%MVF (D) in placebo (PLA, gray) (5 males and 8 females) and quercetin (QUE, green) (6 males and 7 females) groups before (PRE) and after 6 weeks of (POST) intervention. *p < 0.050
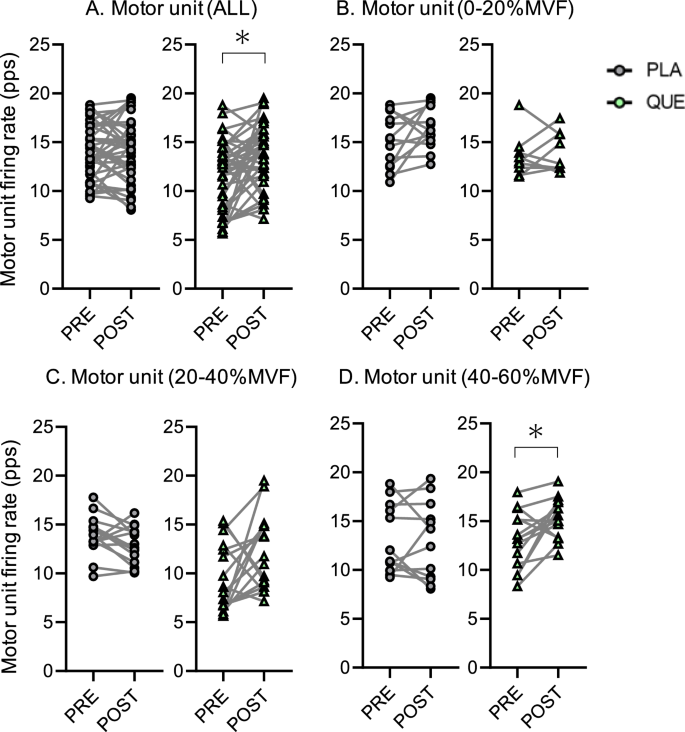
Numbers of tracked MUALL from PRE to POST in PLA and QUE were 39, respectively. There was a significant difference in the firing rate of tracked MUALL between PRE and POST in QUE (p = 0.005, r = 0.28) but not in PLA (p = 0.885, r = 0.11) (Fig. 5A). Numbers of tracked MU0–20 from PRE to POST in PLA and QUE were 13 and 9, respectively. There was no significant difference in the firing rate of tracked MU0–20 between PRE and POST in either group (PLA, p = 0.110, r = 0.18; QUE, p = 0.734, r = 0.09) (Fig. 5B). Numbers of tracked MU20–40 from PRE to POST in PLA and QUE were 14 and 16, respectively. There was no significant difference in the firing rate of tracked MU20–40 between PRE and POST in either group (PLA, p = 0.104, r = 0.14; QUE, p = 0.117, r = 0.20) (Fig. 5C). Numbers of tracked MU40–60 from PRE to POST in PLA and QUE were 12 and 14, respectively. There was a significant difference in the firing rate of tracked MU40–60 between PRE and POST in QUE (p = 0.025, r = 0.33) but not in PLA (p = 0.850, r = 0.12) (Fig. 5D).
Firing rate tracked from PRE to POST of all motor units (A) and motor unit recruitment threshold between 0 and 20%MVF (B), 20 and 40%MVF (C), and 40 and 60%MVF (D) in placebo (PLA, gray) (5 males and 8 females) and quercetin (QUE, green) (6 males and 7 females) groups before (PRE) and after 6 weeks of (POST) intervention. *p < 0.050
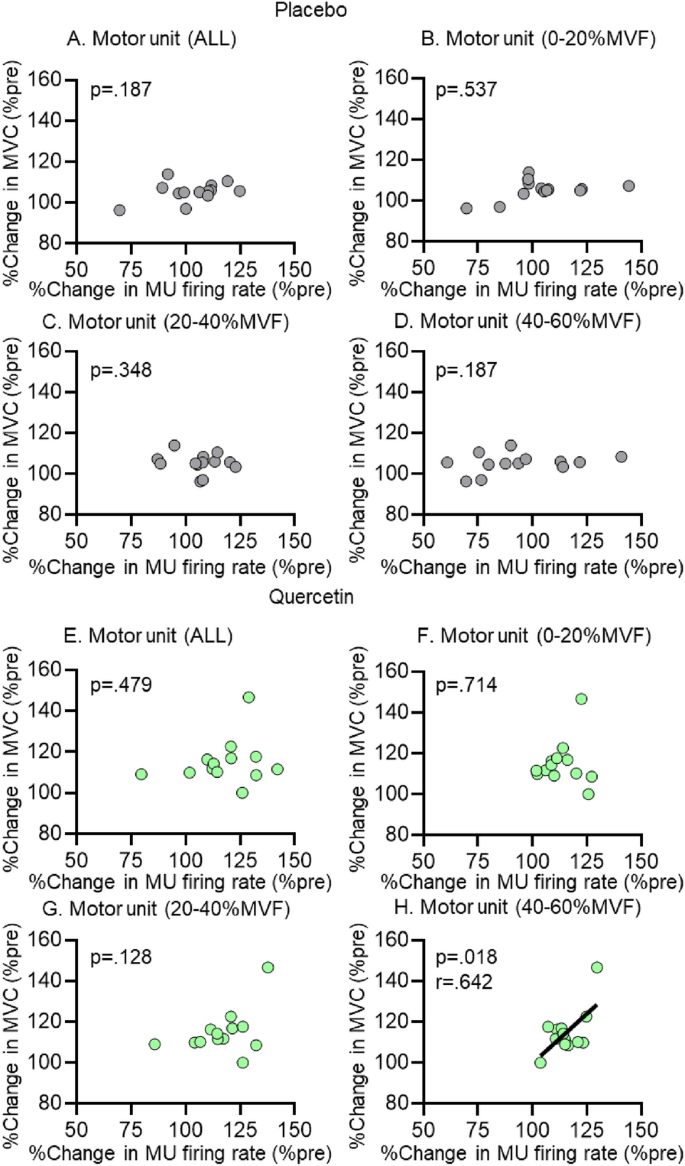
In PLA, there was no significant correlation between %change in MVF and %change in the firing rate of MU0–20, MU20–40, MU40–60, or MUALL from PRE to POST (p > 0.050) (Fig. 6A–D). In QUE, there was a significant correlation between %change in MVF and %change in firing rate of MU40–60 from PRE to POST (p = 0.018, r = 0.642), but not for MU0–20, MU20–40, or MUALL (all, p > 0.050) (Fig. 6E–H).
Pearson correlation between %change in maximal voluntary force (MVF) and %change in firing rate of motor units (MU). The results of all MU (A, E), MU recruitment threshold between 0 and 20%MVF (B, F), 20 and 40%MVF (C, G), and 40 and 60%MVF (D, H) from before and after intervention in placebo (5 males and 8 females) and quercetin (6 males and 7 females) groups are shown. The line shows the linear regression line and indicates significance (p < 0.05)
Discussion
This study examined the effects of quercetin ingestion on MUs behavior during a single session of resistance exercise and on chronic adaptations in muscle strength and MUs behavior following resistance training in older adults. The main findings of the present study were that quercetin ingestion: (1) led to MUs with a relatively higher recruitment threshold exhibiting greater fatigue during a single session of resistance training (Fig. 3D); (2) enhanced the increase in muscle strength following resistance training (Table 2); (3) altered adaptations of MUs with a relatively higher recruitment threshold to resistance training (Figs. 4, 5); and (4) led to an increase in muscle strength associated with a change in MUs with a higher recruitment threshold behavior following resistance training (Fig. 6). These results suggest that quercetin ingestion enhances improvements of muscle strength following resistance training by promoting adaptations of MUs with higher recruitment thresholds in older adults.
Effects of quercetin ingestion on motor unit behavior during a single session of resistance exercise
Most %change in the MU firing rate from the first to the last contractions during a single session of resistance training was generally below 100%, since the MU firing rate decreased due to resistance exercise (Fig. 3). These data indicate that individual MUs had fatigued due to resistance exercise. Changes in the MU firing rate with fatigue have been reported to represent an increase [41, 42], decrease [33, 43,44,45], or no change [46]. Previous studies showed that the individual MU firing rate decreased from the beginning to end of fatiguing exercise [44, 47]. Since the present study analyzed only MUs recruited from the first contraction, declines in the MU firing rate represent fatigue induced by resistance exercise. Although it is difficult to explain the detailed physiological pathways based on our data, it is possible that the MU firing rate was modulated by inhibitory afferent signals from receptors sensitive to either changes in muscle contractile properties or the metabolic state of the muscle [44, 48]. In both groups, the decrease in the firing rate of MU40–60 following exercise was notable from PRE to MID (Fig. 3D). A previous study, in which young adults performed resistance training including ballistic contraction for 4 weeks, reported progressively greater MU conduction velocity for MUs with a higher recruitment threshold [49]. The specific changes in conduction velocity observed in MUs after short-term resistance training might be due to specific adaptations in voltage-sensitive ionic channels (e.g., Na + and K +) and/or modifications of the transport activity and capacity of the Na + –K + pump (e.g., Na + –K + –ATPase) [49]. As mentioned above, the MU firing rate was affected by muscle contractile properties or the metabolic state of muscle [44, 48]. In the present study, the firing rate of MUs with a higher recruitment threshold was increased following 3 weeks of resistance training, and MUs with a higher recruitment threshold exhibited greater fatigue due to a single session of such training. There was a moderate magnitude interaction of %changes in the MU firing rate of MU40–60 (p < 0.05), but not in those of MU0–20 or MU20–40 (p > 0.05) (Fig. 3B–D). These data indicate that quercetin ingestion resulted in a greater increase in fatigue of MU40–60 due to resistance exercise. Martinez-Valdes et al. [50] reported that an overall increase in conduction velocity of MUs with a lower recruitment threshold was observed following 2 weeks of either high-intensity interval training or moderate-intensity continuous training, whereas the conduction velocity of MUs with a higher recruitment threshold increased significantly only with high-intensity interval training. This suggests that activation of MUs with a higher recruitment threshold during training enhances adaptations of MUs with a higher recruitment threshold to the training. We found that quercetin ingestion altered MU activation properties, i.e., enhancements of activation of MUs with a higher recruitment threshold during resistance training, in older adults, as reported in previous studies involving young adults (Nishikawa et al. 2023; Watanabe and Holobar 2021). Quercetin is structurally similar to adenosine; it blocks adenosine binding to A1 and A2a receptors and releases neurotransmitters such as acetylcholine and dopamine [20]. It is possible that the effects of quercetin ingestion on the central, via modulation of synaptic input, affect the MUs recruitment and firing patterns [51]. While this study does not allow for a detailed description of the physiological pathways, one of the mechanisms leading to the recruitment threshold-dependent effect could be that quercetin may potentially act on specific synaptic pathways. MUs with lower recruitment threshold are activated via mono-synaptic pathways, while those recruited with higher recruitment threshold are activated through both mono- and polysynaptic pathways [52]. From these findings, we considered that resistance training in conjunction with quercetin ingestion could selectively activate MUs with a higher recruitment threshold in older adults, and that chronic intervention might also induce different adaptations in MU.
Effects of resistance training and quercetin ingestion
In PLA, MVF was approximately 5% greater (significantly) due to resistance training (Table 2). This improvement of MVF was smaller than in previous studies with similar research designs [32, 37]. The previous studies, in which older adults underwent 6 weeks of isometric knee extension resistance training at 80%MVF, reported increases in MVF of 19% [32] or 14% [37]. Such a smaller increase in muscle strength following intervention may be due to the lower exercise intensity (60% of MVF) applied in the present study, as suggested previously [9, 10]. On the other hand, MVF was about 15% greater (significantly) with the combination of resistance training and quercetin ingestion. These data show that quercetin ingestion improves the increase in MVF by resistance training, supporting our hypothesis. Improves the increase in MVF by resistance training in QUE is large magnitude greater than in PLA. Also, the rate of improvement in muscle strength in QUE (15%) was similar with that in previous studies that used 80%MVF [32, 37], which means that resistance training with moderate intensity plus daily quercetin ingestion would induce similar changes in muscle strength as resistance training at a high intensity in older adults.
For TUGnormal and TUGfast, there was a significant main effect of Time, but no such effect was observed for Chair5times or Chair30sec (Table 2). Additionally, there was no interaction effect observed across all physical fitness tests in this study (Table 2). Our data suggest that quercetin ingestion could not improve increasing physical fitness for walking, standing up, or sitting down but could improve strength increases induced by resistance training. Strength of the knee extensor was one of the factors used to determine the results of physical fitness tests in this study. However, previous studies reported that parameters other than the strength of the knee extensor are associated with the results of physical fitness tests, and so any assessment of the results of such tests would require a more comprehensive approach [53]. A previous study of older adults undergoing 12 weeks of resistance training reported that isometric knee extension strength was increased, but the chair stand test and walking speed results were similar from pre to post resistance training [54]. Further studies are warranted to examine the effects of quercetin ingestion on the outcomes of functional training interventions, not just knee extension resistance training, on the physical fitness of older adults.
In both groups, muscle thickness of the vastus lateralis and vastus intermedius, as well as the muscle volume of the right lower limb were similar from PRE to POST (Table 2). Resistance training is known to enhance neural factors during early periods before the adaptation of peripheral factors, such as muscle hypertrophy [55]. Supporting this, a previous study reported that 6 weeks of low-, moderate-, or high-intensity resistance training led to increases in strength, without a corresponding increase in the muscle cross-sectional area [56]. The duration of the resistance training period in this study may have been too short to cause muscle hypertrophy. Hence, the observed increase in muscle strength resulting from resistance training may be attributed to adaptations in neural factors rather than peripheral factors in the current study.
In both groups, the firing rate of MUALL was similar from PRE to POST (Fig. 4A). Following chronic resistance training, the MU firing rate is generally increased/decreased during submaximal contractions [32, 37, 40]. No changes in the MU firing rate would be caused by the exercise protocols, such as the intensity, used in the present study. A previous study reported that the intensity during resistance training is a key determinant of improvements in efferent neural drive in older adults [57]. There were significant interactions for the firing rate of MUALL, MU20–40, and MU40–60, and MU firing rates at POST were greater in QUE than PLA (Fig. 4A, C, D). Furthermore, the firing rate of MUALL and MU40–60 tracked from PRE to POST was increased from PRE to POST in QUE, but not in PLA (Fig. 5A, D). Our data suggest that quercetin ingestion increased the firing rate of MUs with relatively moderate and higher recruitment thresholds after 6 weeks of resistance training. The effect size results of the present study show that quercetin ingestion has a moderate effect on the firing rate of MU with relatively moderate or higher thresholds before and after resistance training. This indicates that quercetin ingestion has significant physiological effects on motor units, and this does not represent an alpha error due to the analysis of many motor units. In addition, our data showed an increase in muscle strength associated with an increase in the firing rate of MUs with a higher recruitment threshold following intervention in QUE (Fig. 6H). A previous study reported that improvements in muscle strength and neural factors such as voluntary activation following high-intensity resistance training were greater than those following low-intensity resistance training, although muscle hypertrophy was similar between them [58]. This indicates that adaptations in muscle strength and neural factors could be partly explained by recruitments of MUs with higher recruitment thresholds that are recruited during high- but not low-intensity resistance training. The present study showed that the combination of resistance training and quercetin ingestion induced adaptations of MUs with relatively moderate or higher recruitment thresholds (Figs. 4, 5 and 6). This may be one of the reasons for the greater increase in muscle strength in the group performing resistance training in conjunction with quercetin rather than placebo ingestion (Table 2).
This study has some limitations that should be addressed. Further research should be conducted in participants with different characteristics, intervention periods, and training intensities than in this study to clarify the detailed effects of quercetin on resistance training adaptation. A previous study reported an increasing in muscle mass following resistance training in sarcopenic older adults was lower than in healthy older adults [59]. If neuromuscular properties of the participants are different, the effects of quercetin on training adaptation would be different. In the present study, the short training period was a factor without hypertrophy to resistance training. It may be possible to observe the effect of quercetin on muscle hypertrophy by conducting over a longer training period [60]. The intensity of training would be important in the effect of quercetin on training adaptation. Since MUs with higher recruitment threshold can recruited during training even if quercetin is not ingestion, quercetin could not affect adaptation to high intensity resistance training. Furthermore, this study did not evaluate quercetin concentration during the intervention. High quercetin concentrations in plasma could be maintained by regular and frequent quercetin ingestion [61,62,63]. Therefore, we used daily quercetin ingestion over intervention periods to alter motor unit behavior during resistance training by quercetin ingestion. This is an important point in this study, as it is necessary for the concentration of quercetin in the body to reach a level that affects motor unit activity. The dose of quercetin at which it acts as an adenosine antagonist in human body is currently unknown. However, previous studies showed quercetin 200–500 mg single or continuous ingestion alter motor unit behavior or neuromuscular properties [21,22,23, 34, 64]. These previous studies supported the results of this study, which showed that the quercetin dose used in this study could alter motor unit behavior during resistance training. Additionally, using a higher dose of quercetin such as used in previous studies [21, 24, 25, 65, 66] may promote the adaptation to resistance training, because the previous study reported the dose–response relationship of quercetin on the motor unit behavior [64]. Thus, future intervention trials should consider the dose–response relationship of quercetin on adaptation to resistance training. Furthermore, previous studies showed sex differences in motor unit behavior [67,68,69,70]. For example, Lecce et al. reported firing rate of MUs with higher recruitment threshold in males is higher than in females and firing rate of MUs with lower recruitment threshold in females is higher in females than in males in young adults [67]. One of the mechanisms of sex differences in motor unit behavior could be potential influence of sex hormones on the central nervous system [71]. Quercetin acts on the central nervous system and is thought to affect motor unit activity, so the effects of quercetin may be influenced by sex differences. Additionally, no clear evidence exists demonstrating that sex affects quercetin pharmacokinetics [72]. Therefore, sex differences may influence the impact of quercetin on motor unit activity and its potential to enhance training adaptations. In future research, the influence of sex differences on the effects of quercetin in resistance training adaptations should be investigated using a larger sample size that includes both male and female participants.
Conclusion
We investigated the effect of quercetin ingestion on MUs behavior and muscle strength following 6 weeks of resistance training in older adults. Our findings indicate that quercetin ingestion enhances the improvements of muscle strength following resistance training involving adaptations of MU with higher recruitment thresholds in older adults. The present study suggests that quercetin ingestion combining quercetin ingestion with resistance training is an effective strategy for improving muscle strength in older adults.
Data availability
The data presented in this study are available on request from the corresponding author.
References
-
Grimby G (1995) Muscle performance and structure in the elderly as studied cross-sectionally and longitudinally. J Gerontol A Biol Sci Med Sci 50A:17–22. https://doi.org/10.1093/gerona/50a.special_issue.17
-
Aagaard P, Suetta C, Caserotti P, Magnusson SP, Kjaer M (2010) Role of the nervous system in sarcopenia and muscle atrophy with aging: strength training as a countermeasure. Scand J Med Sci Sports 20(1):49–64. https://doi.org/10.1111/j.1600-0838.2009.01084.x
-
Janssen I (2011) The epidemiology of sarcopenia. Clin Geriatr Med 27(3):355–363. https://doi.org/10.1016/j.cger.2011.03.004
-
Beaudart C, Zaaria M, Pasleau F, Reginster JY, Bruyere O (2017) Health outcomes of sarcopenia: a systematic review and meta-analysis. PLoS ONE 12(1):e0169548. https://doi.org/10.1371/journal.pone.0169548
-
Bickel CS, Cross JM, Bamman MM (2011) Exercise dosing to retain resistance training adaptations in young and older adults. Med Sci Sports Exerc 43(7):1177–1187. https://doi.org/10.1249/MSS.0b013e318207c15d
-
Hunter GR, McCarthy JP, Bamman MM (2004) Effects of resistance training on older adults. Sports Med 34(5):329–348. https://doi.org/10.2165/00007256-200434050-00005
-
Hunter GR, Wetzstein CJ, McLafferty CL Jr, Zuckerman PA, Landers KA, Bamman MM (2001) High-resistance versus variable-resistance training in older adults. Med Sci Sports Exerc 33(10):1759–1764. https://doi.org/10.1097/00005768-200110000-00022
-
Vechin FC, Libardi CA, Conceicao MS, Damas FR, Lixandrao ME, Berton RP et al (2015) Comparisons between low-intensity resistance training with blood flow restriction and high-intensity resistance training on quadriceps muscle mass and strength in elderly. J Strength Cond Res 29(4):1071–1076. https://doi.org/10.1519/JSC.0000000000000703
-
Borde R, Hortobagyi T, Granacher U (2015) Dose-response relationships of resistance training in healthy old adults: a systematic review and meta-analysis. Sports Med 45(12):1693–1720. https://doi.org/10.1007/s40279-015-0385-9
-
Steib S, Schoene D, Pfeifer K (2010) Dose-response relationship of resistance training in older adults: a meta-analysis. Med Sci Sports Exerc 42(5):902–914. https://doi.org/10.1249/MSS.0b013e3181c34465
-
Miller JD, Lippman JD, Trevino MA, Herda TJ (2020) Neural drive is greater for a high-intensity contraction than for moderate-intensity contractions performed to fatigue. J Strength Cond Res 34(11):3013–3021. https://doi.org/10.1519/JSC.0000000000003694
-
Henneman E, Somjen G, Carpenter DO (1965) Functional significance of cell size in spinal motoneurons. J Neurophysiol 28:560–580. https://doi.org/10.1152/jn.1965.28.3.560
-
Hug F, Avrillon S, Ibanez J, Farina D (2023) Common synaptic input, synergies and size principle: control of spinal motor neurons for movement generation. J Physiol 601(1):11–20. https://doi.org/10.1113/JP283698
-
Liu CJ, Latham N (2010) Adverse events reported in progressive resistance strength training trials in older adults: 2 sides of a coin. Arch Phys Med Rehabil 91(9):1471–1473. https://doi.org/10.1016/j.apmr.2010.06.001
-
Queiroz AC, Kanegusuku H, Chehuen MR, Costa LA, Wallerstein LF, Dias da Silva VJ et al (2013) Cardiac work remains high after strength exercise in elderly. Int J Sports Med 34(5):391–397. https://doi.org/10.1055/s-0032-1323779
-
Pollock RD, Woledge RC, Martin FC, Newham DJ (2012) Effects of whole body vibration on motor unit recruitment and threshold. J Appl Physiol 112(3):388–395. https://doi.org/10.1152/japplphysiol.01223.2010
-
Fatela P, Mendonca GV, Veloso AP, Avela J, Mil-Homens P (2019) Blood flow restriction alters motor unit behavior during resistance exercise. Int J Sports Med 40(9):555–562. https://doi.org/10.1055/a-0888-8816
-
Machado A, Garcia-Lopez D, Gonzalez-Gallego J, Garatachea N (2010) Whole-body vibration training increases muscle strength and mass in older women: a randomized-controlled trial. Scand J Med Sci Sports 20(2):200–207. https://doi.org/10.1111/j.1600-0838.2009.00919.x
-
Yasuda T, Fukumura K, Uchida Y, Koshi H, Iida H, Masamune K et al (2015) Effects of low-load, elastic band resistance training combined with blood flow restriction on muscle size and arterial stiffness in older adults. J Gerontol A Biol Sci Med Sci 70(8):950–958. https://doi.org/10.1093/gerona/glu084
-
Alexander SP (2006) Flavonoids as antagonists at A1 adenosine receptors. Phytother Res 20(11):1009–1012. https://doi.org/10.1002/ptr.1975
-
Watanabe K, Holobar A (2021) Quercetin ingestion modifies human motor unit firing patterns and muscle contractile properties. Exp Brain Res 239(5):1567–1579. https://doi.org/10.1007/s00221-021-06085-w
-
Nishikawa T, Hirono T, Holobar A, Kunugi S, Okudaira M, Ohya T et al (2024) Acute effects of caffeine or quercetin ingestion on motor unit firing pattern before and after resistance exercise. Eur J Appl Physiol. https://doi.org/10.1007/s00421-023-05376-0
-
Nishikawa T, Hirono T, Takeda R, Okudaira M, Ohya T, Watanabe K (2023) Effects of 7-day quercetin intervention on motor unit activity and muscle contractile properties before and after resistance exercise in young adults randomized controlled trials. Appl Physiol Nutr Metab. https://doi.org/10.1139/apnm-2023-0208
-
Patrizio F, Ditroilo M, Felici F, Duranti G, De Vito G, Sabatini S et al (2018) The acute effect of Quercetin on muscle performance following a single resistance training session. Eur J Appl Physiol 118(5):1021–1031. https://doi.org/10.1007/s00421-018-3834-y
-
Bazzucchi I, Patrizio F, Ceci R, Duranti G, Sgro P, Sabatini S et al (2019) The effects of quercetin supplementation on eccentric exercise-induced muscle damage. Nutrients. https://doi.org/10.3390/nu11010205
-
Murota K, Matsuda N, Kashino Y, Fujikura Y, Nakamura T, Kato Y et al (2010) alpha-Oligoglucosylation of a sugar moiety enhances the bioavailability of quercetin glucosides in humans. Arch Biochem Biophys 501(1):91–97. https://doi.org/10.1016/j.abb.2010.06.036
-
Kobayashi S, Honda S, Murakami K, Sasaki S, Okubo H, Hirota N et al (2012) Both comprehensive and brief self-administered diet history questionnaires satisfactorily rank nutrient intakes in Japanese adults. J Epidemiol 22(2):151–159. https://doi.org/10.2188/jea.je20110075
-
Bazzucchi I, Felici F, Montini M, Figura F, Sacchetti M (2011) Caffeine improves neuromuscular function during maximal dynamic exercise. Muscle Nerve 43(6):839–844. https://doi.org/10.1002/mus.21995
-
Jones CJ, Rikli RE, Beam WC (1999) A 30-s chair-stand test as a measure of lower body strength in community-residing older adults. Res Q Exerc Sport 70(2):113–119. https://doi.org/10.1080/02701367.1999.10608028
-
Holobar A, Farina D, Gazzoni M, Merletti R, Zazula D (2009) Estimating motor unit discharge patterns from high-density surface electromyogram. Clin Neurophysiol 120(3):551–562. https://doi.org/10.1016/j.clinph.2008.10.160
-
Watanabe K, Holobar A, Kouzaki M, Ogawa M, Akima H, Moritani T (2016) Age-related changes in motor unit firing pattern of vastus lateralis muscle during low-moderate contraction. Age (Dordr) 38(3):48. https://doi.org/10.1007/s11357-016-9915-0
-
Watanabe K, Holobar A, Mita Y, Tomita A, Yoshiko A, Kouzaki M et al (2020) Modulation of neural and muscular adaptation processes during resistance training by fish protein ingestions in older adults. J Gerontol A Biol Sci Med Sci 75(5):867–874. https://doi.org/10.1093/gerona/glz215
-
Nishikawa T, Takeda R, Hirono T, Okudaira M, Ohya T, Watanabe K (2024) Differences in acute neuromuscular response after single session of resistance exercise between young and older adults. Exp Gerontol 185:112346. https://doi.org/10.1016/j.exger.2023.112346
-
Nishikawa T, Hirono T, Takeda R, Okudaira M, Ohya T, Watanabe K (2024) One-week quercetin intervention modifies motor unit recruitment patterns before and during resistance exercise in older adults: a randomized controlled trial. Physiol Behav 282:114585. https://doi.org/10.1016/j.physbeh.2024.114585
-
Francic A, Holobar A (2021) On the reuse of motor unit filters in high density surface electromyograms recorded at different contraction levels. IEEE Access 9:115227–115236. https://doi.org/10.1109/Access.2021.3104762
-
Hirono T, Kunugi S, Yoshimura A, Holobar A, Watanabe K (2022) Acute changes in motor unit discharge property after concentric versus eccentric contraction exercise in knee extensor. J Electromyogr Kinesiol 67:102704. https://doi.org/10.1016/j.jelekin.2022.102704
-
Watanabe K, Holobar A, Tomita A, Mita Y (2020) Effect of milk fat globule membrane supplementation on motor unit adaptation following resistance training in older adults. Physiol Rep 8(12):e14491. https://doi.org/10.14814/phy2.14491
-
Goodlich BI, Del Vecchio A, Kavanagh JJ (2023) Motor unit tracking using blind source separation filters and waveform cross-correlations: reliability under physiological and pharmacological conditions. J Appl Physiol 135(2):362–374. https://doi.org/10.1152/japplphysiol.00271.2023
-
Martinez-Valdes E, Negro F, Laine CM, Falla D, Mayer F, Farina D (2017) Tracking motor units longitudinally across experimental sessions with high-density surface electromyography. J Physiol 595(5):1479–1496. https://doi.org/10.1113/JP273662
-
Del Vecchio A, Casolo A, Negro F, Scorcelletti M, Bazzucchi I, Enoka R et al (2019) The increase in muscle force after 4 weeks of strength training is mediated by adaptations in motor unit recruitment and rate coding. J Physiol 597(7):1873–1887. https://doi.org/10.1113/JP277250
-
Contessa P, De Luca CJ, Kline JC (2016) The compensatory interaction between motor unit firing behavior and muscle force during fatigue. J Neurophysiol 116(4):1579–1585. https://doi.org/10.1152/jn.00347.2016
-
Muddle TWD, Colquhoun RJ, Magrini MA, Luera MJ, DeFreitas JM, Jenkins NDM (2018) Effects of fatiguing, submaximal high- versus low-torque isometric exercise on motor unit recruitment and firing behavior. Physiol Rep 6(8):e13675. https://doi.org/10.14814/phy2.13675
-
Rubinstein S, Kamen G (2005) Decreases in motor unit firing rate during sustained maximal-effort contractions in young and older adults. J Electromyogr Kinesiol 15(6):536–543. https://doi.org/10.1016/j.jelekin.2005.04.001
-
McManus L, Hu X, Rymer WZ, Lowery MM, Suresh NL (2015) Changes in motor unit behavior following isometric fatigue of the first dorsal interosseous muscle. J Neurophysiol 113(9):3186–3196. https://doi.org/10.1152/jn.00146.2015
-
Vila-Cha C, Falla D, Correia MV, Farina D (2012) Adjustments in motor unit properties during fatiguing contractions after training. Med Sci Sports Exerc 44(4):616–624. https://doi.org/10.1249/MSS.0b013e318235d81d
-
Christova P, Kossev A (2001) Human motor unit recruitment and derecruitment during long lasting intermittent contractions. J Electromyogr Kinesiol 11(3):189–196. https://doi.org/10.1016/s1050-6411(00)00052-3
-
Carpentier A, Duchateau J, Hainaut K (2001) Motor unit behaviour and contractile changes during fatigue in the human first dorsal interosseus. J Physiol 534(Pt 3):903–912. https://doi.org/10.1111/j.1469-7793.2001.00903.x
-
Bigland-Ritchie BR, Dawson NJ, Johansson RS, Lippold OC (1986) Reflex origin for the slowing of motoneurone firing rates in fatigue of human voluntary contractions. J Physiol 379:451–459. https://doi.org/10.1113/jphysiol.1986.sp016263
-
Casolo A, Farina D, Falla D, Bazzucchi I, Felici F, Del Vecchio A (2020) Strength training increases conduction velocity of high-threshold motor units. Med Sci Sports Exerc 52(4):955–967. https://doi.org/10.1249/MSS.0000000000002196
-
Martinez-Valdes E, Farina D, Negro F, Del Vecchio A, Falla D (2018) Early motor unit conduction velocity changes to high-intensity interval training versus continuous training. Med Sci Sports Exerc 50(11):2339–2350. https://doi.org/10.1249/MSS.0000000000001705
-
ter Haar Romeny BM, Denier van der Gon JJ, Gielen CC (1982) Changes in recruitment order of motor units in the human biceps muscle. Exp Neurol 78(2):360–368. https://doi.org/10.1016/0014-4886(82)90054-1
-
Romaiguere P, Vedel JP, Azulay JP, Pagni S (1991) Differential activation of motor units in the wrist extensor muscles during the tonic vibration reflex in man. J Physiol 444:645–667. https://doi.org/10.1113/jphysiol.1991.sp018899
-
Krebs DE, Scarborough DM, McGibbon CA (2007) Functional vs. strength training in disabled elderly outpatients. Am J Phys Med Rehabil 86(2):93–103. https://doi.org/10.1097/PHM.0b013e31802ede64
-
Skelton DA, Young A, Greig CA, Malbut KE (1995) Effects of resistance training on strength, power, and selected functional abilities of women aged 75 and older. J Am Geriatr Soc 43(10):1081–1087. https://doi.org/10.1111/j.1532-5415.1995.tb07004.x
-
Moritani T, deVries HA (1980) Potential for gross muscle hypertrophy in older men. J Gerontol 35(5):672–682. https://doi.org/10.1093/geronj/35.5.672
-
Lasevicius T, Ugrinowitsch C, Schoenfeld BJ, Roschel H, Tavares LD, De Souza EO et al (2018) Effects of different intensities of resistance training with equated volume load on muscle strength and hypertrophy. Eur J Sport Sci 18(6):772–780. https://doi.org/10.1080/17461391.2018.1450898
-
Unhjem R, Toien T, Kvellestad ACG, Oren TS, Wang E (2021) External resistance is imperative for training-induced efferent neural drive enhancement in older adults. J Gerontol A Biol Sci Med Sci 76(2):224–232. https://doi.org/10.1093/gerona/glaa160
-
Jenkins NDM, Miramonti AA, Hill EC, Smith CM, Cochrane-Snyman KC, Housh TJ et al (2017) Greater neural adaptations following high- vs low-load resistance training. Front Physiol 8:331. https://doi.org/10.3389/fphys.2017.00331
-
Negaresh R, Ranjbar R, Baker JS, Habibi A, Mokhtarzade M, Gharibvand MM et al (2019) Skeletal muscle hypertrophy, insulin-like growth factor 1, myostatin and follistatin in healthy and sarcopenic elderly men: the effect of whole-body resistance training. Int J Prev Med 10:29. https://doi.org/10.4103/ijpvm.IJPVM_310_17
-
Pearcey GEP, Alizedah S, Power KE, Button DC (2021) Chronic resistance training: is it time to rethink the time course of neural contributions to strength gain? Eur J Appl Physiol 121(9):2413–2422. https://doi.org/10.1007/s00421-021-04730-4
-
Manach C, Williamson G, Morand C, Scalbert A, Remesy C (2005) Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am J Clin Nutr 81(1 Suppl):230S-242S. https://doi.org/10.1093/ajcn/81.1.230S
-
Shi Y, Williamson G (2016) Quercetin lowers plasma uric acid in pre-hyperuricaemic males: a randomised, double-blinded, placebo-controlled, cross-over trial. Br J Nutr 115(5):800–806. https://doi.org/10.1017/S0007114515005310
-
Egert S, Wolffram S, Bosy-Westphal A, Boesch-Saadatmandi C, Wagner AE, Frank J et al (2008) Daily quercetin supplementation dose-dependently increases plasma quercetin concentrations in healthy humans. J Nutr 138(9):1615–1621. https://doi.org/10.1093/jn/138.9.1615
-
Watanabe K, Kunugi S, Holobar A (2023) The dose-response relationship of quercetin on the motor unit firing patterns and contractile properties of muscle in men and women. J Int Soc Sports Nutr 20(1):2265140. https://doi.org/10.1080/15502783.2023.2265140
-
Davis JM, Carlstedt CJ, Chen S, Carmichael MD, Murphy EA (2010) The dietary flavonoid quercetin increases VO(2max) and endurance capacity. Int J Sport Nutr Exerc Metab 20(1):56–62. https://doi.org/10.1123/ijsnem.20.1.56
-
Nieman DC, Williams AS, Shanely RA, Jin F, McAnulty SR, Triplett NT et al (2010) Quercetin’s influence on exercise performance and muscle mitochondrial biogenesis. Med Sci Sports Exerc 42(2):338–345. https://doi.org/10.1249/MSS.0b013e3181b18fa3
-
Lecce E, Conti A, Nuccio S, Felici F, Bazzucchi I (2024) Characterising sex-related differences in lower- and higher-threshold motor unit behaviour through high-density surface electromyography. Exp Physiol 109(8):1317–1329. https://doi.org/10.1113/EP091823
-
Guo Y, Jones EJ, Inns TB, Ely IA, Stashuk DW, Wilkinson DJ et al (2022) Neuromuscular recruitment strategies of the vastus lateralis according to sex. Acta Physiol (Oxf) 235(2):e13803. https://doi.org/10.1111/apha.13803
-
Jenz ST, Beauchamp JA, Gomes MM, Negro F, Heckman CJ, Pearcey GEP (2023) Estimates of persistent inward currents in lower limb motoneurons are larger in females than in males. J Neurophysiol 129(6):1322–1333. https://doi.org/10.1152/jn.00043.2023
-
Nishikawa Y, Watanabe K, Holobar A, Kitamura R, Maeda N, Hyngstrom AS (2024) Sex differences in laterality of motor unit firing behavior of the first dorsal interosseous muscle in strength-matched healthy young males and females. Eur J Appl Physiol 124(7):1979–1990. https://doi.org/10.1007/s00421-024-05420-7
-
Piasecki J, Skarabot J, Spillane P, Piasecki M, Ansdell P (2024) Sex differences in neuromuscular aging: the role of sex hormones. Exerc Sport Sci Rev 52(2):54–62. https://doi.org/10.1249/JES.0000000000000335
-
Guo Y, Bruno RS (2015) Endogenous and exogenous mediators of quercetin bioavailability. J Nutr Biochem 26(3):201–210. https://doi.org/10.1016/j.jnutbio.2014.10.008
Acknowledgements
The authors are grateful to Prof. Aleš Holobar of the University of Maribor, Slovenia, for supporting analyses of motor unit firing properties using the DEMUSE tool.
Funding
This study was supported by the Bilateral Program of JSPS (JPJSBP-82626) to KW.
Author information
Authors and Affiliations
-
Graduate School of Health and Sport Sciences, Chukyo University, Toyota, Japan
Taichi Nishikawa
-
Laboratory of Neuromuscular Biomechanics, School of Health and Sport Sciences, Chukyo University, Toyota, Japan
Taichi Nishikawa, Ryosuke Takeda, Saeko Ueda, Kaito Igawa, Tetsuya Hirono, Masamichi Okudaira & Kohei Watanabe
-
Department of Human Nutrition, School of Life Studies, Sugiyama Jogakuen University, Nagoya, Japan
Saeko Ueda & Yukiko Mita
-
Human Health Sciences, Graduate School of Medicine, Kyoto University, Kyoto, Japan
Tetsuya Hirono
-
Faculty of Education, Iwate University, Morioka, Japan
Masamichi Okudaira
-
Laboratory for Exercise Physiology and Biomechanics, Graduate School of Health and Sport Sciences, Chukyo University, Toyota, Japan
Toshiyuki Ohya
Corresponding author
Correspondence to Kohei Watanabe.
Original Author:[Taichi Nishikawa, Ryosuke Takeda, Saeko Ueda, Kaito lgawa, Tetsuya Hirono, MasamichiOkudaira, Yukiko Mita, Toshiyuki Ohya & Kohei Watanabe]
Source:[SPRINGER-https://link.springer.com/article/10.1007/s00394-025-03634-9]
This article is shared for informational and educational purposes only and does not represent the views of our brand. If there are any copyright concerns, please contact us and we will address them promptly.